우수연구성과
Chung-Ang University Researchers Unravel Role of Cohesins In Embryonic Stem Cell Division
Chung-Ang University Researchers
Unravel Role of Cohesins In Embryonic Stem Cell Division
Study shows how meiotic cohesin complexes affect chromosome
structure and function—and the long-term implications of their effects on the stem
cell genome
The eukaryotic chromosomal structure depends on the role
played by complexes of specific proteins called cohesins. Cohesins play an
active role during mitotic and meiotic cell division (which is responsible for
maintaining chromosomal integrity). Now, researchers highlight the
supplementary role of the meiotic cohesin complex in mitotic cell division as
observed in embryonic stem cells. The findings could have far-reaching consequences
on investigations into chromosomal diseases such as Down’s syndrome, certain
cancers, and infertility.
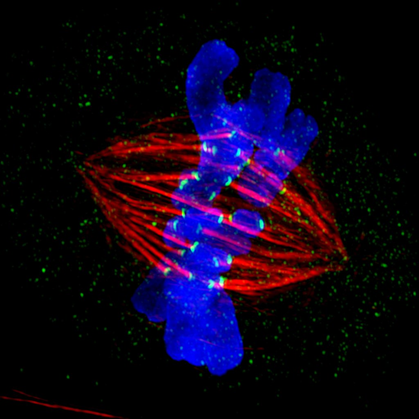
In a new study, Chung-Ang University researchers show for the first time how specific cohesin complexes involved in meiotic cell division affect chromosome structure and function in mitotic cell division.
Image
courtesy: ZEISS Microscopy on Creative Commons
Licensed under CC BY-NC-ND 2.0.
Chromosomes undergo precise structural changes at a molecular level
during the different phases of cell division. These changes occur at a high
level of accuracy to prevent genome instability. Genome instability resulting
from broken, missing, or rearranged chromosomes has been found to be the root
cause of cell death, carcinogenesis, and congenital disorders. Studying genomic
instability helps researchers identify the causes of cancer and may lead to new
advancements in their diagnosis and treatment.
Scientists have long deciphered the role played
by cohesin during meiotic cell division. Cohesins are important proteins for
maintaining genome integrity as they ensure faithful chromosome segregation by
holding sister chromatids together during meiosis. Now, in a new study conducted by scientists
led by Professor Keun P. Kim from Chung-Ang
University, South Korea, embryonic stem cells (ESCs) were studied to understand
whether the meiosis-specific cohesin complex plays an active role during mitotic
cell division, which is responsible for the growth and development of all
cells. Their findings were published online on 3 March 2022 in Genome
Biology.
ESCs show pluripotency or the potential to differentiate into
every cell type in the body, and also have an unlimited capacity for
self-renewal and a high tolerance for DNA damage stress. “However, we know little about how ESCs maintain genome integrity and
cope with the chromosomal abnormalities and replication stresses that can occur
during cell proliferation and differentiation,” explains Prof. Kim. To
bridge this knowledge gap, the team identified two cohesin factors, REC8 and STAG3,
which are specifically expressed in ESCs. They studied the possible
contribution of these meiotic components during mitotic cell divisions that are
closely linked with the structure and organization of chromosomes in ESCs.
Based on the existing information about meiotic cohesins, the team
used ESCs derived from mice to analyze the expression pattern of mitotic
cohesin components. High resolution 3D-SIM (Structured Illumination Microscopy)
and functional analyses were employed to understand how REC8 and STAG3
contributed to chromosome structure and cellular function in mitotic divisions.
The team found that if the amount of cohesin proteins
in the cell is suppressed, the chromosomes exhibit severe compaction, resulting
in their early non-separation—ultimately leading to an unstable genome. To
prevent this crisis, it is necessary to maintain adequate levels of the cohesin
factors REC8 and STAG3, which ensure chromosomal stabilization and adequate
sister chromatid cohesion during the cell cycle in ESCs.
This study proves that chromosomal morphogenesis and
interaction depend upon the presence or absence of mitotic and meiotic cohesin
factors. The findings provide an improved understanding into the process of
chromatid cohesion and chromosome formation in mitotic ESC chromosomes. Commenting
on the applications of their study, Prof. Kim concludes: “The purpose of our study was to provide an answer for how the cohesin
complex and other regulatory factors are involved in the formation of the chromosome
and maintenance of genomic integrity. We think this will be helpful in
researching the mechanisms and treatment methods of diseases like cancer,
infertility, and chromosomal diseases such as Down syndrome.”
Reference
|
Authors
Title of original paper
Journal |
Eui‑Hwan Choi1,
Seobin Yoon1, Young Eun Koh1, Tae Kyung Hong2,
Jeong Tae Do2, Bum‑Kyu Lee3, Yoonsoo Hahn1,
and Keun P. Kim1
Meiosis‑specific cohesin
complexes display essential and distinct roles in mitotic embryonic
stem cell chromosomes
Genome Biology |
|
|
|
|
DOI
Affiliations |
·
1Department of Life Sciences, Chung-Ang University, Seoul, 06974, South
Korea. ·
2Department of Stem Cell and Regenerative Biotechnology, Konkuk
Institute of Technology, Konkuk University, Seoul, 05029, South Korea. ·
3Department of Biomedical Sciences, Cancer Research Center, University
of Albany-State University of New York, Rensselaer, NY, USA.
|
About Chung-Ang University
Chung-Ang University is a private
comprehensive research university located in Seoul, South Korea. It was started
as a kindergarten in 1918 and attained university status in 1953. It is fully
accredited by the Ministry of Education of Korea. Chung-Ang University conducts
research activities under the slogan of “Justice and Truth.” Its new vision for
completing 100 years is “The Global Creative Leader.” Chung-Ang University
offers undergraduate, postgraduate, and doctoral programs, which encompass a
law school, management program, and medical school; it has 16 undergraduate and
graduate schools each. Chung-Ang University’s culture and arts programs are
considered the best in Korea.
Website: https://neweng.cau.ac.kr/index.do
About Professor Keun P. Kim
Professor Keun P. Kim is a Professor of Life Science at Chung-Ang
University. Before coming to Chung-Ang University, he completed his Postdoctoral
training at Nancy Kleckner’s lab at Harvard University. In 2005, Prof. Kim
received a PhD in Molecular Biology from Seoul National University.
The Kim group studies fundamental genetic recombination and
chromosome dynamics using 3D high resolution imaging, physical analysis of
recombination, and molecular genetic approaches to monitor meiosis. The group
is also presently studying DNA repair pathways and homologous recombination:
two processes that are essential for maintaining genomic integrity in diverse
organisms.
